Cellular Biophysics
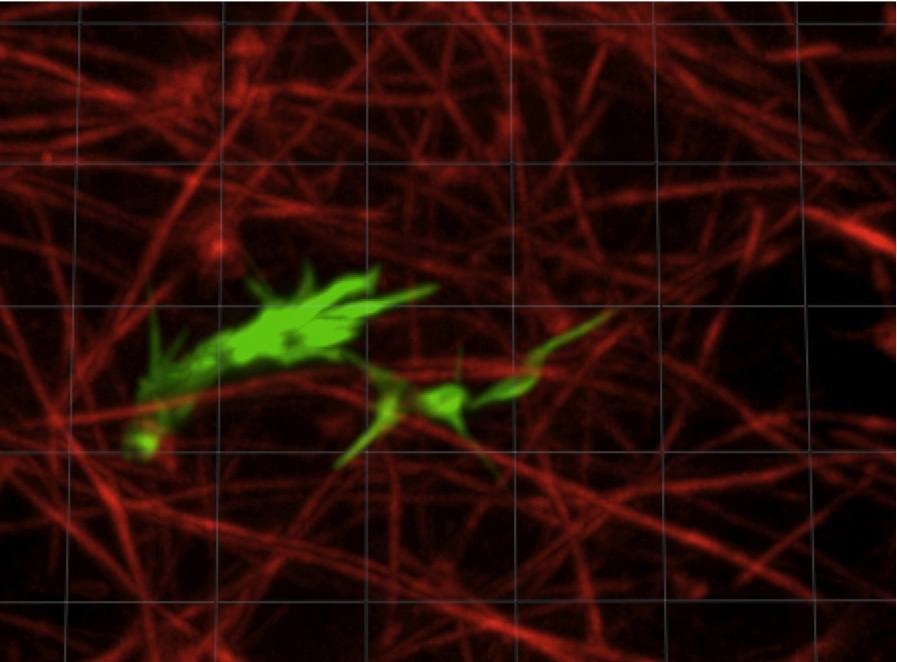 Axon navigating in a 3D collagen gel. The actin from the axon is stained in green, the collagen fibrils in red.
Axon navigating in a 3D collagen gel. The actin from the axon is stained in green, the collagen fibrils in red.
Axon Motility and Guidance
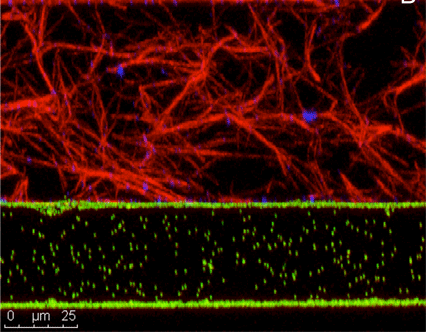
How do axons sense molecular gradients and use these signals to find their correct targets? How does the extracellular matrix modulate axon motility? In collaboration with Herb Geller an the NIH, we are studying the dynamics of axons navigating in collagen matrices. We have developed a novel technology for generating precisely controlled molecular gradients in collagen gels, and are using it to study the behavior of developing and regenerating axons. The growing neurons are led by a complex, dynamic structure called a growth cone, and we are trying to understand how it works as a sensing element. Recently we have begun investing the biomechanics of the axon outgrowth through live cell imaging of neurons transfected with fluorescently labeled cytoskeletal proteins. Funded by NIH. Click on the link for recent publications.
Giardia
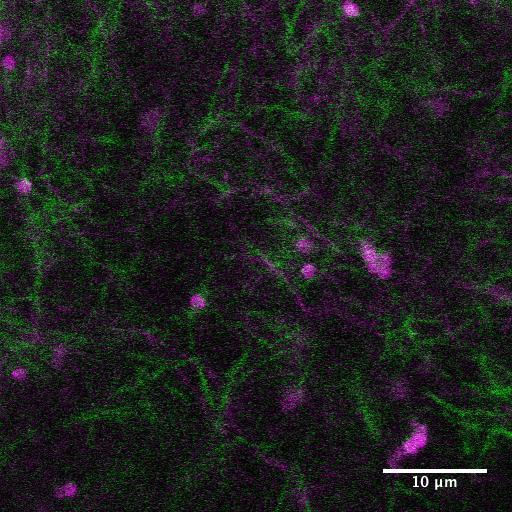
Giardia lamblia is one of the most prevalent intestinal protozoan pathogens worldwide, causing widespread infection in areas without access to clean water. In much of the world, the lack of clean water results in nearly universal infection of the population by the age of three. After ingestion of the infective cyst stage by the host, the parasite differentiates in the lumen of the small intestine into the trophozoite form, attaches to intestinal epithelial cells (the lower intestine) and replicates. Its presence often results in severe gastrointestinal symptoms, including diarrhea, vomiting and weight loss.
In collaboration with Heidi Elmendorf’s lab, we are helping to answer questions about Giardia’s cytoskeleton and how attaches itself to the intestinal wall. Ongoing experiments are helping to determine the mechanism of attachment employed by Giardia which is believed to be, for the most part, due either to a suction mechanism or to a gripping mechanism. Funded by NIH.
Living P. falciparum parasite inside a human red blood cell
Malaria
In collaboration with Professor Paul Roepe and his group, we are imaging the structure and dynamics of the malarial parasite after invasion of a red blood cell. Using a high speed, high resolution confocal microscope, we have investigated the volume regulation of intracellular compartments in order to understand the mechanism of drug resistance. Funding from NIH.